Abstract
Introduction Adolescent and young adult (AYA) patients (pts) with acute lymphoblastic leukemia (ALL) have favorable outcomes when treated with pediatric inspired ALL regimens. Pediatric ALL regimens rely on asparaginase and corticosteroids and thus are associated with metabolic toxicities including hepatotoxicity, pancreatitis, hyperglycemia and lipid disorders, which may be exacerbated by obesity. Obesity has been associated with inferior overall survival (OS) among children and AYA pts with ALL (Butturini JCO 2007; Stock Blood 2019). In this study we investigate the association of body mass index (BMI) with metabolic toxicities and survival among AYA pts with ALL treated-on Dana-Farber Cancer Institute (DFCI) ALL Consortium regimens.
Methods All pts aged 15-50 years treated on four sequential multi-center DFCI ALL Consortium protocols were included: Pediatric 00-001, Pediatric 05-001, Adult 01-175, and Adult 06-254. Additional consecutive pts treated as per these protocols at DFCI, Massachusetts General Hospital, and Boston Children's Hospital were included (2008-2021) with data extracted from the electronic medical record. Body mass Index (BMI) was calculated based on CDC guidelines - per age-adjusted percentiles up to age 20 years (underweight: < 5%; normal: 5-84.99%; overweight: 85-94.99%; obese: ≥95%) and per absolute BMI (kg/m2) in age ≥ 20 years (underweight: < 18.5; normal: 18.5-24.99; overweight 25-29.99; and obese ≥ 30). For this analysis, underweight and normal categories were unified. Toxicities were determined by CTCAE 2.0,3.0 and 5.0 with hepatotoxicity defined as grade III/IV elevated ALT, AST or total bilirubin, and dyslipidemia defined as grade III/IV hypertriglyceridemia or hypercholesterolemia.
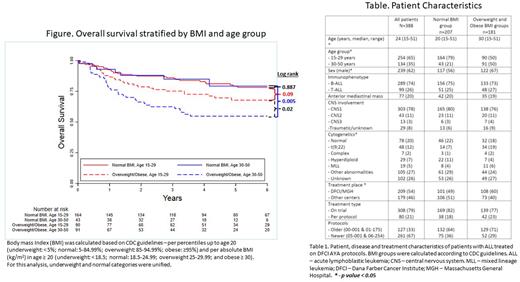
Results In total 388 pts were included with a median age of 24 (range 15-50) years. BMI at diagnosis was normal in 207 (53.3%) and overweight/obese in 181 (46.6%) pts (Table). With a median follow up of 5.5 years, the 4-year OS was 74%, higher in younger (15-29 years) vs older (30-50 years) AYA pts (79% vs 64%, p=0.003), and higher in pts with normal vs overweight/obese BMI (83% vs 64%, p=0.001). Younger vs older AYAs more commonly had normal BMI (79% vs 20%, p<0.0001), thus we conducted a stratified analysis. In pts with normal BMI, the 4-year OS was similarly excellent between younger vs older AYAs (83% vs 85%, p=0.89). In contrast, in overweight/obese pts, the 4-year OS was higher in younger versus older AYAs (73% vs 55%, p=0.02; Figure). Older AYA obese pts (n=39) had a particularly poor 4-year OS of 47%vs 72% among younger obese AYAs (p=0.019).
The 4-year event free survival (EFS) was higher in normal vs overweight/obese pts (77% vs 63%, p=0.003). In pts with normal BMI the 4-year EFS was similar between younger vs older AYAs (78% vs 74%, p=0.63), whereas in obese pts the 4-year EFS was higher in younger vs older AYAs (72 vs 49%, p=0.07). In a cox multivariable analysis, both immunophenotype (B- vs T-ALL) and BMI (overweight/obese vs normal) were independently associated with inferior OS (HR 2.11, CI 95% 1.23-3.60, p=0.006, and HR 2.15, CI 95% 1.45-3.19, p<0.001, respectively), whereas age was not. In a sensitivity analysis excluding pts with Ph positive ALL (n=48), BMI and ALL immunophenotype retained their prognostic value.
Regarding toxicity, grade III/IV hepatotoxicity and hyperglycemia rates were lower in pts with normal BMI vs overweight/obese pts (37% vs 58%, p<0.001 and 21 vs 35%, p=0.003, respectively). Hyperlipidemia and pancreatitis rates were not statistically different comparing BMI groups (28% vs 21%, p=0.11 and 13% in both groups, p=0.92, respectively). When stratified by age, within younger AYAs, more hepatotoxicity, hyperglycemia and hyperlipidemia were seen in overweight/obese vs. normal BMI pts (53% vs 32%, p< 0.001; 33% vs 19%, p=0.01 and 31 vs 20%, p=0.05, respectively). In contrast, there was no significant association between BMI and toxicity in older AYAs.
Conclusion Among pts aged 15-50 years treated with DFCI pediatric regimen, similar excellent outcomes were seen among younger and older AYAs with normal BMI. Overweight and obese patients fared poorly, particularly in the older group where markedly inferior OS and EFS was detected despite equivalent rates of metabolic toxicities, suggesting obesity may promote disease resistance and require additional novel approaches.
Disclosures
Place:Novartis: Consultancy, Other: Meal, Research Funding; Abbvie: Consultancy, Research Funding; Servier: Consultancy, Research Funding. Silverman:Jazz Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Servier: Membership on an entity's Board of Directors or advisory committees. Brunner:GSK: Research Funding; Celgene/BMS: Consultancy, Research Funding; Keros Therapeutics: Consultancy; Takeda: Consultancy, Research Funding; Acceleron: Honoraria; Agios: Honoraria; Taiho: Consultancy; Novartis: Consultancy, Research Funding; AstraZeneca: Research Funding; Janssen: Research Funding; Aprea: Research Funding. Stone:Syntrix: Consultancy; Syndax: Consultancy; OncoNova: Consultancy; Novartis: Consultancy; Kura Oncology: Consultancy; Jazz: Consultancy; Janssen: Consultancy; Innate: Consultancy; GSK: Consultancy; Gemoab: Consultancy; Foghorn Therapeutics: Consultancy; Epizyme: Consultancy; Elevate Bio: Consultancy; BMS: Consultancy; Boston Pharmaceuticals: Consultancy; BerGenBio: Consultancy; Astellas: Consultancy; Arog: Consultancy, Research Funding; Apteva: Consultancy; Aprea: Consultancy; Actinium: Consultancy; Abbvie: Consultancy, Research Funding; Takeda: Consultancy; Syros: Consultancy. Neuberg:Madrigal Pharmaceuticals: Current equity holder in private company. DeAngelo:Abbvie: Research Funding; Forty-Seven: Consultancy; Autolus: Consultancy; Agios: Consultancy; Blueprint Medicines Corporation: Consultancy; Incyte: Consultancy; Amgen: Consultancy; Glycomimetics: Research Funding; Shire: Consultancy; Takeda: Consultancy; Novartis: Consultancy, Research Funding; Pfizer: Consultancy; Jazz Pharmaceuticals: Consultancy. Luskin:Pfizer: Honoraria; Abbvie: Research Funding; Novartis: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal